Thrombotic Thrombocytopenia after ChAdOx1 nCov-19 Vaccination
List of authors.

Andreas Greinacher Awarded the Vox Sanguinis Best Paper
- Andreas Greinacher, M.D.,
- Thomas Thiele, M.D.,
- Theodore E. Warkentin, M.D.,
- Karin Weisser, Ph.D.,
- Paul A. Kyrle, M.D.,
- and Sabine Eichinger, M.D.
Metrics
Abstract
Background
Several cases of unusual thrombotic events and thrombocytopenia have developed after vaccination with the recombinant adenoviral vector encoding the spike protein antigen of severe acute respiratory syndrome coronavirus 2 (SARS-CoV-2) (ChAdOx1 nCov-19, AstraZeneca). More data were needed on the pathogenesis of this unusual clotting disorder.
Methods
We assessed the clinical and laboratory features of 11 patients in Germany and Austria in whom thrombosis or thrombocytopenia had developed after vaccination with ChAdOx1 nCov-19. We used a standard enzyme-linked immunosorbent assay to detect platelet factor 4 (PF4)–heparin antibodies and a modified (PF4-enhanced) platelet-activation test to detect platelet-activating antibodies under various reaction conditions. Included in this testing were samples from patients who had blood samples referred for investigation of vaccine-associated thrombotic events, with 28 testing positive on a screening PF4–heparin immunoassay.
Results
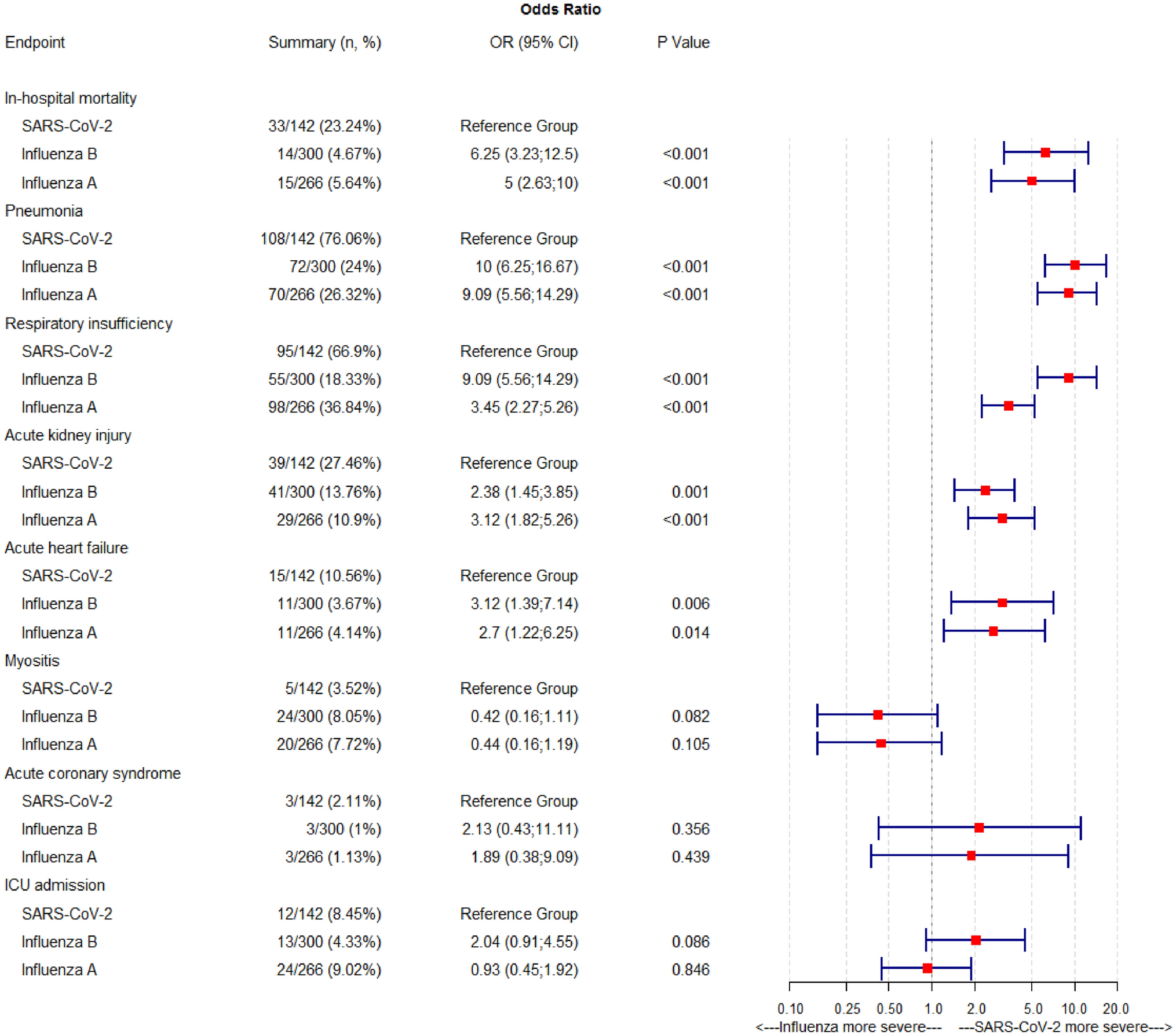
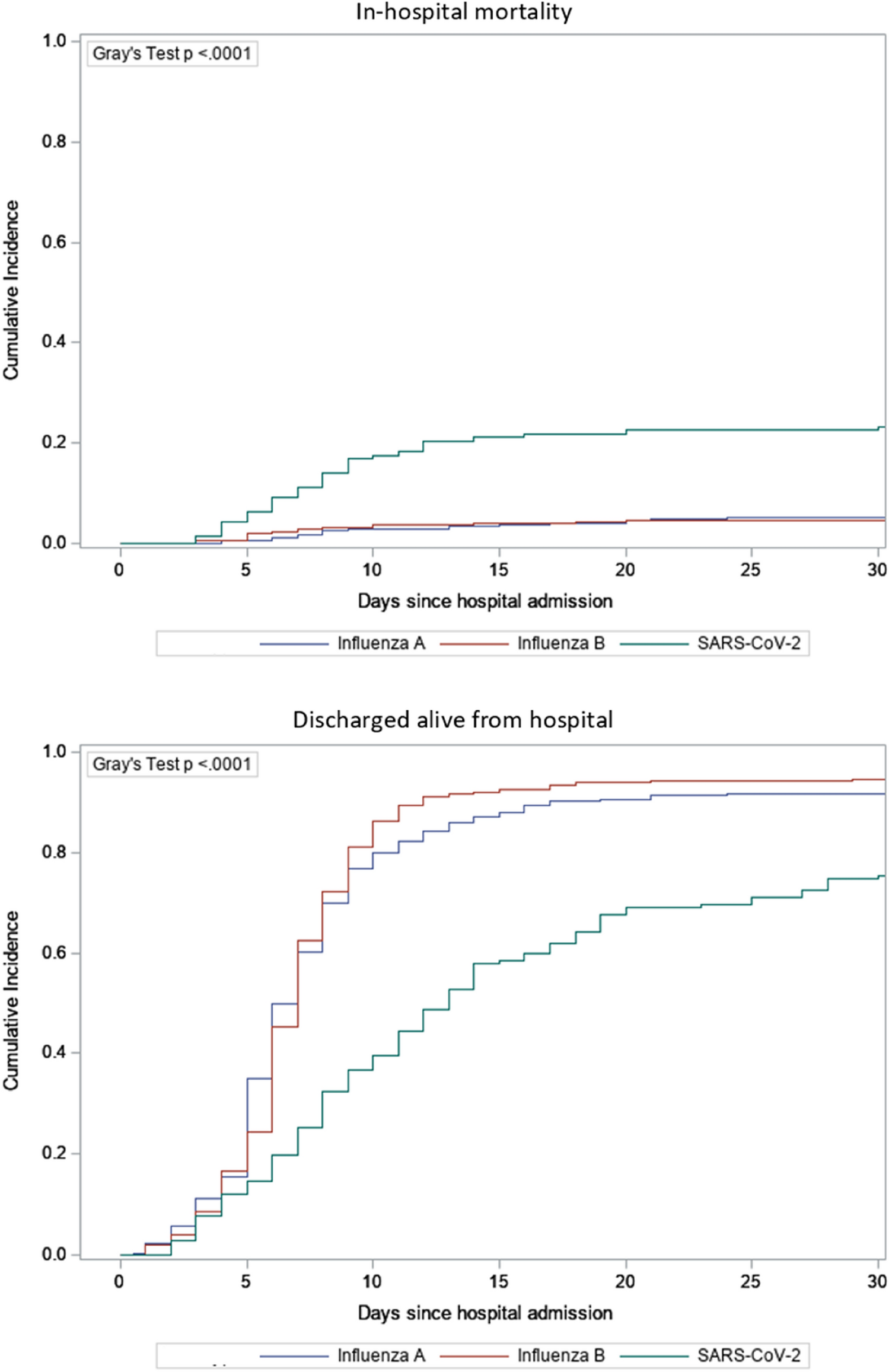
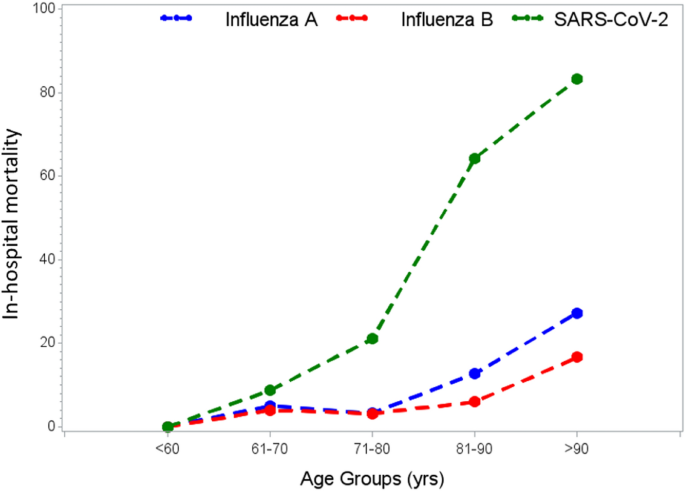
Of the 11 original patients, 9 were women, with a median age of 36 years (range, 22 to 49). Beginning 5 to 16 days after vaccination, the patients presented with one or more thrombotic events, with the exception of 1 patient, who presented with fatal intracranial hemorrhage. Of the patients with one or more thrombotic events, 9 had cerebral venous thrombosis, 3 had splanchnic-vein thrombosis, 3 had pulmonary embolism, and 4 had other thromboses; of these patients, 6 died. Five patients had disseminated intravascular coagulation. None of the patients had received heparin before symptom onset. All 28 patients who tested positive for antibodies against PF4–heparin tested positive on the platelet-activation assay in the presence of PF4 independent of heparin. Platelet activation was inhibited by high levels of heparin, Fc receptor–blocking monoclonal antibody, and immune globulin (10 mg per milliliter). Additional studies with PF4 or PF4–heparin affinity purified antibodies in 2 patients confirmed PF4-dependent platelet activation.
Conclusions
Vaccination with ChAdOx1 nCov-19 can result in the rare development of immune thrombotic thrombocytopenia mediated by platelet-activating antibodies against PF4, which clinically mimics autoimmune heparin-induced thrombocytopenia. (Funded by the German Research Foundation.)
Vaccines against severe acute respiratory syndrome coronavirus 2 (SARS-CoV-2) are the most important countermeasure to fight the coronavirus 2019 (Covid-19) pandemic. From December 2020 through March 2021, the European Medicines Agency approved four vaccines on the basis of randomized, blinded, controlled trials: two messenger RNA–based vaccines — BNT162b2 (Pfizer–BioNTech) and mRNA-1273 (Moderna) — that encode the spike protein antigen of SARS-CoV-2, encapsulated in lipid nanoparticles; ChAdOx1 nCov-19 (AstraZeneca), a recombinant chimpanzee adenoviral vector encoding the spike glycoprotein of SARS-CoV-2; and Ad26.COV2.S (Johnson & Johnson/Janssen), a recombinant adenovirus type 26 vector encoding SARS-CoV-2 spike glycoprotein.
As of April 7, 2021, more than 82 million vaccine doses had been administered in the European Union; in Germany, approximately one quarter of vaccine recipients had received the ChAdOx1 nCov-19 vaccine.1 Beginning in late February 2021, several cases of unusual thrombotic events in combination with thrombocytopenia were observed in patients after vaccination with ChAdOx1 nCov-19.
Index Case
A previously healthy 49-year-old health care worker received her first dose of ChAdOx1 nCov-19 in mid-February 2021 (day 0). Over the next few days, she reported having minor symptoms (fatigue, myalgia, and headache). Beginning on day 5, she reported having chills, fever, nausea, and epigastric discomfort; she was admitted to a local hospital on day 10.
Table 1.
Laboratory Characteristics of the Index Patient.
Laboratory results are shown in Table 1. The platelet count was 18,000 per cubic millimeter, and the d-dimer level was 35 mg per liter (reference value, <0.5). The results of other blood tests were normal except for γ-glutamyltransferase and C-reactive protein levels, which were elevated. SARS-CoV-2 reverse-transcriptase–polymerase-chain-reaction assay of a nasopharyngeal swab was negative.
Computed tomography (CT) showed portal-vein thrombosis and peripheral pulmonary emboli. The patient received a platelet concentrate and was transferred to a tertiary hospital. On arrival, she had epigastric discomfort and nausea but was otherwise in good condition (blood pressure, 125/88 mm Hg; heart rate, 65 beats per minute; temperature, 36.5°C). The physical examination was unremarkable except for moderate epigastric pain on palpation. She received intravenous antibiotics, analgesia, and one 4000-unit dose of low-molecular-weight heparin (enoxaparin), given subcutaneously.
The following day, the platelet count and fibrinogen level remained low, and the d-dimer and aminotransferase levels increased. The abdominal pain worsened, and repeat CT imaging showed progression of portal-vein thrombosis to include the splenic and upper mesenteric veins; in addition, small thrombi were visualized in the infrarenal aorta and both iliac arteries. Low-dose intravenous unfractionated heparin (500 IU per hour) was initiated but was stopped shortly thereafter because of a sudden onset of tachycardia and concern for gastrointestinal bleeding. The lactate level was 3.7 mmol per liter, and the patient was transferred to the intensive care unit. Repeat CT imaging revealed diffuse gastrointestinal bleeding with reduced perfusion of the intestinal wall and pancreas by splanchnic-vein thrombosis, along with ascites. She received red-cell and platelet transfusions, prothrombin complex concentrates, and recombinant factor VIIa but died on day 11. In addition to the diagnosed medical findings, autopsy revealed cerebral venous thrombosis.
Case Series
Table 2. Clinical and Laboratory Summary of 11 Patients with Available Clinical Information.
By March 15, 2021, an additional 10 patients for whom clinical data were available were found to have one or more thrombotic complications beginning 5 to 16 days after vaccination with ChAdOx1 nCov-19. Characteristics of all 11 patients (including the index case) are presented in Table 2. Thrombotic events included cerebral venous thrombosis (in 9 patients), splanchnic-vein thrombosis (in 3 patients), pulmonary embolism (in 3 patients), and other types of thrombi (in 4 patients); 5 of 10 patients had more than one thrombotic event. Included in this analysis is one patient (Patient 11) who presented with fatal cerebral hemorrhage. The results of brain neuropathological analysis were pending at the time of this report, and cerebral venous thrombosis had not been ruled out; postmortem serum was available for testing for platelet-activating antibodies.
Among these patients, the median age was 36 years (range, 22 to 49); 9 of 11 were women. All the patients presented with concomitant thrombocytopenia (median nadir of platelet count, approximately 20,000 per cubic millimeter; range, 9000 to 107,000). One patient had preexisting von Willebrand disease, anticardiolipin antibodies, and factor V Leiden. None of the patients had received heparin before the onset of symptoms or the diagnosis of thrombosis. Given the striking clinical resemblance of this disorder to autoimmune heparin-induced thrombocytopenia (a prothrombotic thrombocytopenic disorder that can be triggered by heparin and certain other anions and that features heparin-independent platelet-activating properties), serum obtained from 4 of the 11 patients was referred for immediate investigation of platelet-activating antibodies directed against platelet factor 4 (PF4)–heparin. After characterizing the antibodies in serum obtained from Patients 1 through 4, we subsequently obtained serum from 5 of the 7 remaining patients. In addition, our reference laboratory received further serum samples from patients who were suspected of having prothrombotic thrombocytopenia related to ChAdOx1 nCov-19 vaccination. (No detailed clinical information regarding these patients was available at the time of this report.)
Methods
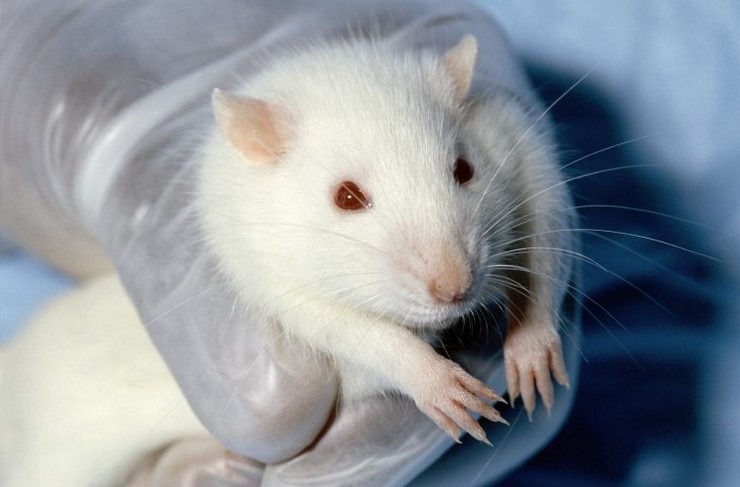
We purified platelets from whole blood (obtained from healthy volunteers) that had undergone anticoagulation with adenine citrate dextrose solution A. None of the volunteers had been taking antiplatelet drugs or had been vaccinated in the previous 10 days. We prepared platelets using methods that have been described previously.2,3 In a subgroup of experiments, platelets were preincubated in buffer with ChAdOx1 nCov-19 (1:2000 dilution) and washed before use. Washed platelets (75 microliters) were incubated with either buffer, a low-molecular-weight heparin (reviparin [Abbott]), or PF4 (Chromatec) in either the presence or absence of the FcγIIa receptor–blocking antibody IV.3. In some experiments, unfractionated heparin (100 IU per milliliter) was added to inhibit PF4-dependent reactions, or ChAdOx1 nCov-19 (1:50 dilution) was added per well. Serum was coincubated with PF4 and platelets in the presence of immune globulin (Privigen IVIG [CSL Behring]) at a concentration of 10 mg per milliliter. After establishing assay conditions using serum from the initial four patients, we investigated another 24 serum samples that tested positive on immunoassay to validate our findings. We refer to this modified platelet-activation test as the PF4-enhanced platelet-activation test.
To measure direct antibody binding, we used two immunoassays, a PF4–heparin enzyme-linked immunosorbent assay (ELISA) and a PF4 ELISA, with antibody binding measured by a secondary antihuman IgG, as described previously.4 In addition, antibodies from two serum samples were affinity purified by immobilized PF4–heparin and immobilized PF4, and the purified antibodies were tested in the assays. (Details about this method are provided in the Supplementary Appendix, available with the full text of this article at NEJM.org.)
We defined reactivity on ELISA according to the optical-density units as strong (≥2.00), intermediate (1.00 to 1.99), or weak (0.50 to 0.99). On the PF4-enhanced platelet-activation test, reactivity was graded according to the time that had elapsed until platelet aggregation,5 with shorter reaction times indicating stronger platelet activation (strong activation, 1 to 5 minutes; intermediate activation, >5 to 15 minutes; and weak activation, >15 to 30 minutes).
Results
All 11 patients in the initial analysis had moderate-to-severe thrombocytopenia and unusual thrombosis, particularly cerebral venous thrombosis and splanchnic-vein thrombosis (Table 2). We also found evidence of disseminated intravascular coagulation in 5 of the patients on the basis of the combination of greatly elevated d-dimer levels (>10.0 mg per liter) and one or more abnormalities in the international normalized ratio, partial thromboplastin time, or fibrinogen level. (Of the 6 patients with available fibrinogen levels, 4 had hypofibrinogenemia.)
Although evaluating the outcomes of different management strategies was not the goal of our study, we noted with interest the clinical course of Patient 2, who presented with pulmonary embolism and mild thrombocytopenia (platelet count, 107,000 per cubic millimeter), without disseminated intravascular coagulation. This patient received therapeutic-dose low-molecular-weight heparin for 3 days, with clinical improvement and an increase in the platelet count to 132,000; at that time, a positive result on PF4–heparin ELISA was obtained, and the patient was switched to oral apixaban, with continued clinical and laboratory recovery.
Figure 1. Reactivity of Patient Serum on Platelet-Activation Assays and Immunoassays.
Table 2 also shows results of the PF4–heparin ELISA, including for the first 4 patients in whom detailed laboratory studies were performed. Serum obtained from these patients showed strong reactivity on PF4–heparin ELISA, with optical densities of more than 3.00 units (reference value, <0.50); all reactivity reactions were inhibited to less than 0.50 units by the addition of heparin (100 IU per milliliter). Figure 1 shows the serologic profile of the 4 initial patients, as assessed by means of the platelet-activation assay. Three of the four serum samples showed weak-to-moderate reactivity at buffer control, which was inhibited by low-molecular-weight heparin. In three of the samples, PF4 (10 μg per milliliter) greatly enhanced reactivity; serum from Patient 2 subsequently showed strong platelet activation in the presence of PF4 when retested along with platelets from other volunteers. All reactions were blocked by monoclonal antibody IV.3 and immune globulin at a dose of 10 mg per milliliter, which indicated that platelet activation had occurred through platelet Fcγ receptors (Figure 1A). None of the controls showed platelet activation (data not shown).
Platelet activation was enhanced when platelets were pelleted from platelet-rich plasma, resuspended in washing buffer, preincubated (1:2000) with ChAdOx1 nCov-19, centrifuged, and resuspended in the final suspension buffer or when they were coincubated in the suspension buffer with ChAdOx1 nCov-19 (1:50). The monoclonal antibody IV.3 blocked PF4-dependent platelet activation in all 7 samples that were tested.
Figure 1B shows the results of platelet activation in serum samples obtained from 24 patients with clinically suspected vaccine-induced immune thrombotic thrombocytopenia who tested positive on the screening PF4–heparin ELISA. Whereas approximately half the serum samples (13 of 24) showed platelet activation at buffer control, most samples (19 of 24) were inhibited by low-molecular-weight heparin; almost all samples (22 of 24) showed platelet activation by the addition of PF4. All but one serum sample was inhibited by a high dose of heparin.
Figure 1C shows strong reactivity of the serum samples obtained from all 28 patients (including Patients 1, 2, 3, 4, 5, 8, 9, 10, and 11) in results on both PF4–heparin and PF4 ELISA, with inhibition by high heparin doses. Antibodies that were affinity purified with the use of either immobilized PF4 or immobilized PF4–heparin showed the same reactivity pattern as the original serum — in other words, they strongly activated platelets in the presence of 10 μg per milliliter of PF4, an effect that was completely inhibited by a high concentration of heparin.
Discussion
The clinical picture of moderate-to-severe thrombocytopenia and thrombotic complications at unusual sites beginning approximately 1 to 2 weeks after vaccination against SARS-CoV-2 with ChAdOx1 nCov-19 suggests a disorder that clinically resembles severe heparin-induced thrombocytopenia, a well-known prothrombotic disorder caused by platelet-activating antibodies that recognize multimolecular complexes between cationic PF4 and anionic heparin.6 However, unlike the usual situation in heparin-induced thrombocytopenia, these vaccinated patients did not receive any heparin to explain the subsequent occurrence of thrombosis and thrombocytopenia.
In recent years, it has been recognized that triggers other than heparin can cause a prothrombotic disorder that strongly resembles heparin-induced thrombocytopenia on both clinical and serologic grounds, including certain polyanionic drugs (e.g., pentosan polysulfate,7 antiangiogenic agent PI-88,8 and hypersulfated chondroitin sulfate8). Such a prothrombotic syndrome has also been observed in the absence of preceding exposure to any polyanionic medication, such as after both viral and bacterial infections9,10 and knee-replacement surgery.11,12 These various clinical scenarios with apparent nonpharmacologic triggers have been classified under the term autoimmune heparin-induced thrombocytopenia.13 Unlike patients with classic heparin-induced thrombocytopenia, patients with autoimmune heparin-induced thrombocytopenia have unusually severe thrombocytopenia, an increased frequency of disseminated intravascular coagulation, and atypical thrombotic events. Serum from these patients strongly activate platelets in the presence of heparin (0.1 to 1.0 IU per milliliter) but also in the absence of heparin (heparin-independent platelet activation). When these unusual antibodies are observed in patients who have thrombocytopenia without preceding heparin exposure, the term “spontaneous” heparin-induced thrombocytopenia syndrome13,14 has been used. Sometimes, patients in whom heparin-induced thrombocytopenia develops after exposure to heparin present with atypical clinical features, such as an onset of thrombocytopenia beginning several days after stopping heparin (delayed-onset heparin-induced thrombocytopenia15,16) or thrombocytopenia that persists for several weeks despite the discontinuation of heparin (persisting or refractory heparin-induced thrombocytopenia17,18). Serum from these patients also shows the phenomenon of heparin-independent platelet-activating properties.
These clinical features that resemble those of autoimmune heparin-induced thrombocytopenia were observed in the patients with vaccine-induced immune thrombotic thrombocytopenia. The serum usually showed strong reactivity on the PF4–heparin ELISA. Moreover, serum showed variable degrees of platelet activation in the presence of buffer that was in most cases greatly enhanced in the presence of PF4 (Figure 1A and 1B). More strikingly, most serum showed inhibition, rather than increased activation, in the presence of low-dose low-molecular-weight heparin (0.2 U per milliliter of anti–factor Xa). In addition, antibodies from two patients, which were affinity purified on either immobilized PF4 or immobilized PF4–heparin, strongly activated platelets but only in the presence of PF4. Enhancement of platelet activation by PF4 is also a feature of heparin-induced thrombocytopenia19,20 and has been used to enhance detection of platelet-activating antibodies in diagnostic testing for this adverse drug reaction.21 Whether these antibodies are autoantibodies against PF4 induced by the strong inflammatory stimulus of vaccination or antibodies induced by the vaccine that cross-react with PF4 and platelets requires further study.
Although we found enhanced reactivity of patient serum with platelets in the presence of ChAdOx1 nCov-19, this is likely to be an in vitro artifact. It is well known that adenovirus binds to platelets22 and causes platelet activation.22,23 Furthermore, the amount of adenovirus in a 500-microliter vaccine injection administered 1 or 2 weeks earlier would seem unlikely to contribute to subsequent platelet activation observed in these patients. However, interactions between the vaccine and platelets or between the vaccine and PF4 could play a role in pathogenesis. One possible trigger of these PF4-reactive antibodies could be free DNA in the vaccine. We have previously shown that DNA and RNA form multimolecular complexes with PF4, which bind antibodies from patients with heparin-induced thrombocytopenia and also induce antibodies against PF4–heparin in a murine model.24 Unfortunately, other Covid-19 vaccines were not available to us for testing.
Our findings have several important clinical implications. First, clinicians should be aware that in some patients, venous or arterial thrombosis can develop at unusual sites such as the brain or abdomen, which becomes clinically apparent approximately 5 to 20 days after vaccination. If such a reaction is accompanied by thrombocytopenia, it can represent an adverse effect of the preceding Covid-19 vaccination. To date, this reaction has been reported only with the ChAdOx1 nCov-19 vaccine, which has been used in approximately 25% of vaccine recipients in Germany and in 30% of those in Austria.
Second, ELISA to detect PF4–heparin antibodies in patients with heparin-induced thrombocytopenia is widely available and can be used to investigate patients for potential postvaccination thrombocytopenia or thrombosis associated with antibodies against PF4.25 A strongly positive ELISA result that is obtained in a patient who has not been recently exposed to heparin would be a striking abnormality.
Third, we have shown that these antibodies recognize PF4 and that the addition of PF4 greatly enhances their detectability in a platelet-activation assay. Since vaccination of millions of persons will be complicated by a background of thrombotic events unrelated to vaccination, a PF4-dependent ELISA or a PF4-enhanced platelet-activation assay may be used to confirm the diagnosis of vaccine-induced immune thrombotic thrombocytopenia through this novel mechanism of postvaccination formation of platelet-activating antibodies against PF4. Although treatment decisions such as administering intravenous immune globulin and starting anticoagulation do not need to await laboratory diagnosis, detection of these unusual platelet-activating antibodies will be highly relevant for case identification and future risk–benefit assessment of this and other vaccines.
Figure 2.
Potential Diagnostic and Therapeutic Strategies for Management of Suspected Vaccine-Induced Immune Thrombotic Thrombocytopenia.
Figure 2 shows a potential diagnostic and therapeutic strategy for managing this novel prothrombotic thrombocytopenic disorder. One consideration is to administer high-dose intravenous immune globulin to inhibit Fcγ receptor–mediated platelet activation. This recommendation parallels emerging experience in the treatment of severe autoimmune heparin-induced thrombocytopenia in which high-dose intravenous immune globulin has resulted in rapid increases in platelet count and de-escalation of hypercoagulability.12,26 We found that the addition of immune globulin in doses that are readily achieved clinically was effective in inhibiting platelet activation by patients’ antibodies. Clinician reluctance to start anticoagulation may be tempered by administering high-dose intravenous immune globulin to raise the platelet count, especially when a patient presents with severe thrombocytopenia and thrombosis, such as cerebral venous thrombosis.
Given the parallels with autoimmune heparin-induced thrombocytopenia, anticoagulant options should include nonheparin anticoagulants used for the management of heparin-induced thrombocytopenia,27 unless a functional test has excluded heparin-dependent enhancement of platelet activation. Finally, we suggest naming this novel entity vaccine-induced immune thrombotic thrombocytopenia (VITT) to avoid confusion with heparin-induced thrombocytopenia.
Supported by a grant (374031971–TRR 240) from the German Research Foundation.
Disclosure forms provided by the authors are available with the full text of this article at NEJM.org.
Drs. Greinacher and Thiele contributed equally to this article.
This article was published on April 9, 2021, at NEJM.org.
We thank the colleagues who provided serum samples for this study, including Dr. Michael Hirschl of Landesklinikum Zwettl, Austria; Dr. Johannes Thaler of the Medical University of Vienna; Dr. Brigitte Keller-Stanislawski, Dr. Dirk Mentzer, and Prof. Dr. Klaus Cichutek of the Paul-Ehrlich-Institut, Langen, Germany; and Prof. Dr. Hans-Georg Bone, Dr. Juliane Alfes, and Dr. Hans-Christian Atzpodien of Klinikum Vest, Recklinghausen; technologists Ulrike Strobel, Carmen Freyer, Katrin Stein, Ines Warnig, and Ricarda Raschke of Transfusion Medicine Greifswald; and the members of the council of the Gesellschaft für Thrombose und Hämostaseforschung (Prof. Dr. Johannes Oldenburg, Dr. Robert Klamroth, Prof. Dr. Florian Langer, and Prof. Dr. Bernd Pötzsch) for their support.
Author Affiliations
From Institut für Immunologie und Transfusionsmedizin, Universitätsmedizin Greifswald, Greifswald (A.G., T.T.), and the Division of Safety of Medicinal Products and Medical Devices, Paul-Ehrlich-Institut (Federal Institute for Vaccines and Biomedicines), Langen (K.W.) — both in Germany; the Departments of Pathology and Molecular Medicine and of Medicine, McMaster University, Hamilton, ON, Canada (T.E.W.); and the Department of Medicine I, Division of Hematology and Hemostaseology, Medical University of Vienna, Vienna (P.A.K., S.E.).
Address reprint requests to Dr. Greinacher at Institut für Immunologie und Transfusionsmedizin, Abteilung Transfusionsmedizin, Sauerbruchstrasse, 17487 Greifswald, Germany.
https://www.nejm.org/doi/10.1056/NEJMoa2104840
NEJ means New England Journal of Medicine





ΌΝΤΩΣ ,ΤΟΥΣ ΕΙΧΑΝ ΠΑΡΑΣΥΡΕΙ ΣΤΟ ΝΕΟ ΣΥΣΤΗΜΑ ,ΤΟΥ ΚΟΜΜΟΥΝΙΣΜΟΥ.
ΤΑΧΑ ΜΟΥ Ο ΕΡΓΆΤΗΣ ,ΤΑΧΑ ΜΟΥ Ο ΑΓΡΟΤΗΣ ΚΑΙ ΟΛΑ ΤΑ ΓΝΩΣΤΑ.
ΑΠΟ ΠΙΣΩ :Τ Α Ε Ρ Π Ε Τ Α.
ΠΙΣΩ ΑΠΟ ΤΗΝ ΚΟΥΡΤΙΝΑ ,Ο ΤΣΑΧΠΙΝΗΣ ΔΡΑΚΟΣ ΡΟΤΣΙΛΝΤ.
Ο Π.Ι.Ν.Δ.Α.Ρ.Ο.Σ
ΚΩΔΙΚΗ ΟΝΟΜΑΣΙΑ.
ΧΑΖΑΡΟΠΡΑΜΑ Ο ΧΙΤΛΕΡΑΚΟΣ.
ΒΟΥΝΤΟΥ ΜΕΣΑ ΣΤΑ ΔΑΣΗ ΟΙ ΝΑΖΙΣΤΕΣ.
ΣΕΛΗΝΗ ΚΑΙ ΤΑ ΣΥΝΑΦΗ.
ΣΕΞΟ ΤΕΤΟΙΑ ,ΞΕΒΡΑΚΩΜΑΤΑ , ΕΠΙΚΛΉΣΕΙΣ ΚΑΙ ΤΑ ΤΟΙΑΥΤΑ.
ΟΛΟΙ ΤΟΥΣ.
ΕΒΡΑΙΟΣ Ο ΧΙΤΛΕΡΑΚΟΣ ΚΑΙ ΑΠΟ ΔΥΟ ΓΟΝΕΙΣ.
ΣΙΓΑ !.
ΑΜΑ ΑΥΤΟΣ ΗΤΑΝ ΙΟΥΔΑΙΟΣ ,ΕΓΩ ΕΙΜΑΙ Ο ΦΟΥ ΜΑΝ ΤΣΟΥ.
“ΝΑΖΙΣΜΟΣ ,ΜΥΣΤΙΚΗ ΕΤΑΙΡΕΙΑ ”
ΒΡΕΙΤΕ ΤΟ ΒΙΒΛΙΟ.
ΠΑΠΑΡΑΣ ΗΤΑΝ .ΕΝΑΣ ΠΑΠΑΡΑΣ ΚΑΙ ΜΙΣΟΣ.
ΑΥΤΟΣ ΟΜΩΣ ΤΟΥΣ ΕΚΑΝΕ ,ΑΥΤΟΝ ΒΑΛΑΝΕ.
ΑΛΛΟΙ ΚΟΥΛΑΝΤΡΙΖΑΝ ΑΠΟ ΠΙΣΩ.
ΤΟ ΧΡΗΜΑ ΔΕΝ ΤΟ ΕΙΧΑΝ ΟΙ ΦΤΩΧΟΜΠΙΝΕΔΕΣ ΙΟΥΔΑΙΟΙ.
ΤΟ ΕΙΧΑΝ ΟΙ ΧΑΖΑΡΟ ΑΙΜΑΤΟΙ.
ΙΟΥΔΑΙΟΙ ΠΛΕΟΝ ΣΤΟ ΘΡΗΣΚΕΥΜΑ.
ΣΑΝ ΤΟΥΣ ΧΑΜΑΙΛΕΟΝΤΕΣ ,ΟΠΟΥ ΕΜΕΝΑΝ ,ΑΥΤΗΣ ΤΗΣ ΠΕΡΙΟΧΗΣ ΘΡΗΣΚΕΥΜΑ ΑΚΟΛΟΥΘΟΥΣΑΝ.
ΑΥΤΟ ΤΟΥΣ ΕΝΔΙΕΦΕΡΕ ;ΤΟ ΘΡΗΣΚΕΥΜΑ ;
ΚΑΘΟΛΟΥ.
ΕΙΝΑΙ ΧΡΙΣΤΙΑΝΟΣ Ο ΜΠΙ ΣΚΑΤΑ ΚΙΣ ;
ΕΙΝΑΙ ΧΡΙΣΤΙΑΝΟΣ Ο ΑΛ 6;
Ο ΣΗΜΙΤΗΣ ;
Ο ΜΠΕΝΑΚ ;
Ο ΤΖΕΦΡΥ ;
ΠΟΣΟΙ ΚΑΙ ΠΟΣΟΙ ΑΛΛΟΙ.
ΑΝΤΕ ,ΒΑΖΑΝΕ ΚΑΙ ΚΑΝΑ ΟΡΙΤΖΙΝΑΛ ΟΒΡΙΟ -ΙΟΥΔΑΙΟ ΣΤΑ ΔΙΚΑ ΤΟΥΣ ,ΤΟΝ ΤΑΙΖΑΝΕ ΚΑΛΑ ,ΒΛΕΠΕ ΡΟΚΦΕΛΕΡ ΚΑΙ ΤΟΝ ΕΙΧΑΝ ΠΙΑ ΣΤΟ ΧΕΡΙ.
Ο ΓΕΡΜΑΝΙΚΟΣ ΣΤΡΆΤΟΣ ,ΕΙΧΕ ΜΈΣΑ ΠΟΛΛΟΥΣ ΕΒΡΑΙΟΥΣ.
ΣΤΡΑΤΙΩΤΕΣ ,ΑΞΙΩΜΑΤΙΚΟΥΣ.
Ο ΟΔΗΓΟΣ ΤΟΥ ΧΙΤΛΕΡ ΗΤΑΝ ΕΒΡΑΙΟΣ.
ΠΡΟΣΠΑΘΗΣΕ ΝΑ ΤΟΥΣ ΠΕΤΑΞΗ ΕΞΩ ΑΠΟ ΤΙΣ ΤΡΑΠΕΖΕΣ ΚΑΙ ΠΟΛΥΕΘΝΙΚΕΣ.
ΕΙΝΑΙ ΑΛΗΘΕΙΑ ΟΜΩΣ ΑΥΤΟ ;
Ή ΚΑΠΟΙΑ ΕΓΙΝΑΝ ΓΙΑ ΤΑ ΜΑΤΙΑ ΤΟΥ ΚΟΣΜΟΥ.
ΞΕΡΟΥΜΕ ,ΟΤ ΠΙΣΩ ΚΑΙ ΑΠΟ ΤΟΥΣ ΔΥΟ ΠΑΓΚΟΣΜΙΟΥΣ ,ΗΤΑΝ ΤΑ ΕΡΠΕΤΑ.
ΜΕΧΡΙ ΤΟ 2016 ,ΕΙΧΕ ΝΙΚΕΣ Η ΓΕΡΜΑΝΊΑ.
ΜΕΤΑ ,ΛΕΝΕ ,ΟΙ ΕΒΡΑΙΟΙ ΚΑΤΕΔΩΣΑΝ ΜΥΣΤΙΚΑ ΣΤΟΥΣ ΑΛΛΟΥΣ ΧΑΖΑΡΟ ΕΒΡΑΙΟΥΣ ,ΤΗΣ ΑΓΓΛΙΑΣ.
ΜΑΛΙΣΤΑ.
Ο ΧΙΤΛΕΡ ,ΕΙΧΕ ΠΕΙ ΟΤΙ ΑΥΤΗ ΗΤΑΝ Η ΑΙΤΙΑ ΠΟΥ ΈΧΑΣΑΝ ΣΤΟΝ ΠΡΩΤΟ.
ΠΡΟΦΑΝΩΣ ,ΟΤΑΝ ΤΟΝ ΧΩΣΑΝ ΣΤΗΝ ΜΥΗΣΗ …ΠΗΡΕ ΤΑ ΕΠΑΝΩ ΤΟΥ ΤΟ ΧΑΖΑΡΙΚΟ ΑΙΜΑ.
ΕΤΣΙ ,ΠΗΡΕ Η ΜΠΑΛΑ ΠΟΛΛΟΥΣ.
ΝΑ ΑΦΗΣΗ ΤΑ ΠΟΥΣΤΙΚΑ Ο ΧΙΤΛΕΡΑΚΟΣ ,ΓΙΑΤΙ ΑΝΤΕΣΤΡΕΨΕ ΟΛΑ ΤΑ ΕΛΛΗΝΙΚΑ ΣΎΜΒΟΛΑ ΚΑΙ ΤΑ ΜΑΥΡΙΣΕ ΚΙΟΛΑΣ.
ΠΟΙΟΣ ΤΟΥ ΤΟ ΕΙΠΕ ΑΥΤΟ ,ΤΟΥ ΑΓΡΑΜΜΑΤΟΥ ΔΕΚΑΝΕΑ ;
ΕΓΩ ;
ΚΑΙ ΓΙΑ ΝΑ ΞΕΡΟΥΜΕ ΑΛΛΟ ΕΝΑ :
Ο ΧΙΤΛΕΡ ,ΔΕΝ ΗΘΕΛΕ ΤΟΝ ΠΟΛΕΜΟ.
ΚΑΤΙ Ή ΚΑΠΟΙΟΙ ΟΜΩΣ ,ΤΟΝ ΕΣΠΡΩΧΝΑΝ ΠΡΟΣ ΤΑ ΕΚΕΙ.
ΕΙΠΑΜΕ :ΗΤΑΝ ΟΡΤΙΝΑΝΤΖΑ.
Ο ΕΥΦΥΕΣΤΑΤΟΣ ΛΕΝΙΝ…ΧΑΖΑΡΟ ΠΑΝΟΥΡΓΟΣ.
ΛΕΦΤΑ ΑΠΟ ΤΙΣ ΧΑΖΑΡΙΚΕΣ ΤΡΑΠΕΖΕΣ.
ΣΤΗΝ ΚΑΘΟΜΙΛΟΥΜΕΝΗ ,ΕΒΡΑΙΟΙ.
ΤΗΝ ΤΡΩΓΑΝ ΣΤΟΝ Κ@ΛΟ ΟΙ ΙΟΥΔΑΙΟΙ ΚΑΙ ΟΙ ΧΑΖΑΡΟΙ ,ΣΤΗΝ ΑΠΟΞΩ.
ΓΙΑ ΘΥΜΗΘΕΙΤΕ ΣΤΗΝ ΟΥ ΧΑΝ .
ΞΕΡΕΤΕ ΠΟΙΟΙ ΣΤΗΣΑΝ ΤΟ ΣΚΗΝΙΚΟ ;
ΑΠΟ ΜΕΣΑ ΑΠΟ ΤΟ ΚΟΜΜΟΥΝΙΣΤΙΚΟ ΚΟΜΜΑ.
ΔΗΛΑΔΗ ;
ΤΑ ΕΡΠΕΤΑ ,ΑΠΟ ΠΙΣΩ .ΔΡΑΚΟΙ.
ΤΟΥΣ ΕΝΟΙΑΞΕ ΕΑΝ ΓΚΡΕΜΙΣΑΝ ΚΑΤΑΓΗΣ ΧΙΛΙΑΔΕΣ ΚΙΝΕΖΟΥΣ ;
ΟΧΙ.
ΕΙΝΑΙ ΟΛΟΙ ΓΙΑ ΜΑΣΑΜΠΟΥΚΑ.
ΞΕΡΩ ΕΒΡΑΙΟΥΣ ,ΠΟΥ ΔΕΝ ΗΤΑΝ ΠΛΟΥΣΙΟΙ.
ΕΒΡΑΙΟΙ…ΟΤΙ ΕΙΝΑΙ.ΙΟΥΔΑΙΟΙ.
ΠΩΣ ΓΙΝΕΤΑΙ ΑΥΤΟΙ ΝΑ ΜΗΝ ΕΙΝΑΙ ΣΤΗΝ ΠΟΛΙΤΙΚΗ ΚΑΙ ΣΤΗΝ ΜΑΣΑΜΠΟΥΚΑ ,ΣΤΙΣ ΤΡΑΠΕΖΕΣ ;
ΑΡΑ ,ΚΑΤΙ ΤΡΕΧΕΙ.
ΕΙΝΑΙ ΕΒΡΑΙΟΣ Ο ΜΠΙ ΣΚΑΤΑ ΚΙΣ ;
ΟΧΙ.
ΧΑΖΑΡΟΣ.
ΔΙΑΒΑΣΤΕ ΚΑΙ ΓΙΑ ΧΑΖΑΡΟΥΣ ,ΚΙΜΜΕΡΙΟΥΣ -ΣΙΚΑΜΒΡΟΥΣ.
ΔΙΑΒΑΣΤΕ ΚΙΜΜΕΡΙΟΙ ΣΥΚΑΜΒΡΟΙ.
ΠΑΝΑΓΙΩΤΗΣ ΛΕΝΤΖΟΣ Ο ΣΥΓΓΡΑΦΈΑΣ.ΔΙΚΗΓΟΡΟΣ.
ΦΙΔΟ ΑΙΜΑΤΑ.
ΑΝΑΚΑΤΩΣΟΥΡΕΣ ,ΔΟΛΟΠΛΟΚΟΙ ,ΜΗΧΑΝΟΡΡΑΦΟΙ ΚΟΠΤΟΓΑΖΩΤΡΙΕΣ !.
ΣΕ ΤΡΩΝΕ ΛΆΧΑΝΟ ΚΑΙ ΜΠΑΜΠΕΣΙΚΑ ΑΒΛΕΠΕΙ.
ΦΟΥΛ ΘΗΡΙΩΔΙΑ.
ΟΜΟΙΩΣ ,ΟΙ ΜΟΓΓΟΛΟΙ.
ΜΗΝ ΤΟ ΨΑΧΝΕΤΕ.ΤΩΡΑ ΕΙΝΑΙ ΔΙΑΣΠΑΡΤΟΙ ΠΑΝΤΟΥ.
ΤΙΓΚΑ ΣΤΟ ΕΡΠΕΤΙΚΟ ΑΙΜΑ Ο ΠΛΑΝΗΤΗΣ.
Ο ΚΥΡΙΟΣ ΣΤΑΛΙΝΑΚΟΣ ;
ΜΟΛΙΣ ΜΠΟΥΚΑΡΑΝ ΤΑ ΓΕΡΜΑΝΙΚΑ ΣΤΡΑΤΕΥΑΜΑΤΑ ΚΑΙ ΠΡΟΗΛΑΥΝΑΝ ,
ΕΧΩ ΔΙΑΒΑΣΕΙ ΣΤΡΑΤΙΩΤΙΚΗ ΙΣΤΟΡΙΑ.
ΜΟΥ ΑΡΕΣΕΙ ΠΟΛΥ.
ΤΟΥΣ ΚΑΘΑΡΙΖΕ ΤΟΥΣ ΑΞΙΟΥΣ ΣΤΡΑΤΙΩΤΙΚΟΥΣ ,ΣΑΝ ΣΤΡΑΓΑΛΙΑ.
Ο ΤΥΠΑΚΟΣ ,ΠΟΥ ΚΑΤΙ ΗΛΙΘΙΟΙ ΕΔΩ ,ΤΟΝ ΕΧΟΥΝ ΚΑΝΕΙ ΘΕΟ.
ΤΟΝ ΑΧΥΡΑΝΘΡΩΠΟ ΤΟΥ ΡΟΤΣΙΛΝΤ.
ΑΡΙΣΤΟΜΑΧΟΣ.